We celebrate 15 years of ISO 13485 in 2025.


ISO 13485 Certified 15+ Years in a Row | World's First & Largest Custom SUT Library
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.
Discover our North American designed and manufactured products.
Experience our Class 7 Clean Room—7 Rooms across 4 locations.
We have Class 7 & 8 molding capabilities.
Explore our cGMP Control for single-use manufacturing.
We celebrate 15 years of ISO 13485.

Due to the fact that safety and quality are non-negotiable in the medical devices industry, regulatory requirements are increasingly stringent throughout every step of a product’s life cycle, including service and delivery. Increasingly, organizations in the industry are expected to demonstrate their quality management processes and ensure best practice in everything they do.
ISO 13485, Medical devices – Quality management systems – Requirements for regulatory purposes, is an internationally agreed standard that sets out the requirements for a quality management system specific to the medical devices industry. It has recently been revised, with the new version published in March 2016.
Foxx Bioprocess products are widely used across industries that require sterile fluid handling and contamination-free workflows. These include biotechnology, pharmaceutical manufacturing, vaccine development, clinical research laboratories, and life sciences research institutions. Single-use systems, filtration components, and bottle assemblies from Foxx help these organizations maintain product integrity and regulatory compliance while improving operational efficiency in both research and production environments.
Yes. FoxxBioprocessprovidescustom single-use assemblies and made-to-order bioprocess systemstailored to the needs of research laboratories and biopharmaceutical manufacturers. Customers can select specific components such as bottle types, tubing materials, connectors, and filtration elements to build a customized system. These flexible configurations helpoptimizefluid transfer processes and improve workflow efficiency in specialized applications such as biologics development, vaccine manufacturing, and cell culture research.
VersaCap® is a patented cap system developed by Foxx Life Sciences that allows multiple fluid connections on laboratory bottles and carboys. It provides leak-proof sealing, interchangeable connectors, and secure tubing integration, making it ideal for sterile fluid transfer applications. VersaCap® technology helps laboratories create closed systems that reduce contamination risks and simplify fluid management processes. This technology is commonly used in Foxx bottle assemblies and carboy systems for bioprocess and laboratory workflows.
VersaCap® is a patented cap system developed by Foxx Life Sciences that allows multiple fluid connections on laboratory bottles and carboys. It provides leak-proof sealing, interchangeable connectors, and secure tubing integration, making it ideal for sterile fluid transfer applications. VersaCap® technology helps laboratories create closed systems that reduce contamination risks and simplify fluid management processes. This technology is commonly used in Foxx bottle assemblies and carboy systems for bioprocess and laboratory workflows.
Foxx Life Sciences manufactures specialized carboys such as EZBio® Titanium Round carboys andEZGrip® carboys designed for safe liquid storage and transfer. These containers are widely used in laboratories and bioprocess facilities to handle media, reagents, and waste solutions. Features such as ergonomic handles, secure closures, and compatibility with VersaCap® systems allow easy integration with tubing and filtration components. This design improves fluid handling efficiency and ensures reliable containment in research and manufacturing environments.
Foxx single-use bottle assemblies are integrated systems that combine laboratory bottles, tubing, connectors, and filters to enable sterile fluid storage and transfer. These assemblies are commonly used for media preparation, buffer storage, and sampling applications in bioprocess workflows. Foxx offers customizable bottle assemblies that allow customers to select the bottle size, tubing type, and connector configuration to match specific process requirements. These solutions help laboratories establish closed fluid paths and reduce contamination risks.
Biotechnology and pharmaceutical companies increasingly prefer single-use technology because it improves operational efficiency and reduces manufacturing complexity. Disposable systems remove the need for cleaning and sterilization, which lowers operational costs and minimizes the risk of cross-contamination between batches. They also allow manufacturers to quickly switch between different products or production volumes without major equipment changes. This flexibility is especially valuable in biologics development, vaccine production, and personalized medicine manufacturing. As the demand for advanced therapies grows, single-use technologies continue to play a major role in modern biopharmaceutical manufacturing.
Yes, Foxx Life Science’s single-use bioprocess systems are designed specifically for Good Manufacturing Practice (GMP) compliant environments used in pharmaceutical and biotechnology production. These systems are manufactured using high-quality materials that meet regulatory requirements such USP Class VI standards. Foxx’s Single-use components are typically gamma-sterilized and packaged to maintain sterility until use. Because they eliminate the need for cleaning validation and reduce cross-contamination risks, single-use technologies are widely used in GMP manufacturing facilities for biologics, vaccines, and advanced therapies. Proper documentation and traceability also support regulatory compliance during audits.
Bioprocessing is generally divided into upstream processing and downstream processing, each requiring specialized equipment. Upstream processing focuses on growing cells or microorganisms and typically uses bioreactors, media preparation systems, tubing assemblies, and sterile filtration components. Downstream processing involves the purification and recovery of biological products and may include filtration systems, chromatography components, storage containers, and sterile transfer systems. Modern bioprocess facilities increasingly rely on single-use technologies to simplify operations and reduce contamination risks. Using the right equipment in both stages ensures efficient production of biologics, vaccines, and therapeutic proteins. Foxx Life Sciences provides single-use systems built to support every stage of your bio process from upstream development to downstream production.
Choosing the right bioprocess tubing and connectors depends on several factors includingfluid compatibility, pressure requirements, sterility level, temperature tolerance, and regulatory compliance. In biopharmaceutical environments, tubing and connectors mustmaintainsterile fluid transfer without leaks or contamination. Materials such assilicone, C-Flex®, and thermoplastic elastomersare commonly used due to their flexibility and chemical resistance. It is also important to select connectors thatallowsecure and quick assembly during process setup.FoxxBioProcessprovides tubing solutions made from materials likesilicone, thermoplastic elastomers (TPE), and fluoropolymers, ensuring chemical compatibility and flexibility.Foxx offers a range of connector technologies such asEZBio®Linksterile connectors, CPC MPC couplings, Y-connectors, and T-connectors, which allow quick and reliable connection or disconnection of fluid lines without compromising sterility.
Sterile fluid transfer is essential in bioprocessing because it prevents contamination during the movement of liquids such as media, buffers, and biologics between vessels. Proper sterile transfer systems useclosed connections, sterile filters, tubing assemblies, and aseptic connectorstomaintaina contamination-free environment. This approach improves product quality, ensures batch consistency, and supports compliance with pharmaceutical manufacturing regulations. Sterile transfer solutions are widely used inupstream cell culture, downstream purification, and final formulation processes. Bymaintainingsterility throughout the workflow, biopharmaceutical manufacturers can reduce product loss, improve process reliability, andmaintainstrict quality standards.
Sterile connectors are critical components in bioprocess fluid transfer because they enableaseptic connections between tubing, containers, and process equipment without introducing contamination. These connectors are designed tomaintaina closed system during fluid transfer, ensuring the sterility of media, buffers, and biologic products. They are commonly used incell culture processes, vaccine manufacturing, and biologics production, where contamination can lead to batch failure. High-quality sterile connectors improve process reliability, simplify system assembly, and support compliance with regulatory standards used in pharmaceutical manufacturing environments.FoxxBioProcessoffers connector solutions such asEZBio®Linksterile connectors and CPC couplingsthat enable secure and reliable connections in single-use fluid transfer systems.
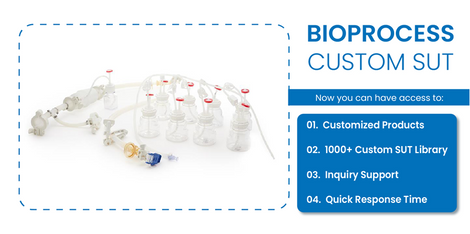
Yes. FoxxBioprocessprovidescustom single-use assemblies and made-to-order bioprocess systemstailored to the needs of research laboratories and biopharmaceutical manufacturers. Customers can select specific components such as bottle types, tubing materials, connectors, and filtration elements to build a customized system. These flexible configurations helpoptimizefluid transfer processes and improve workflow efficiency in specialized applications such as biologics development, vaccine manufacturing, and cell culture research.Foxx Bioprocess Custom SUTis an all-in-one solution for Bioprocess Industryandprovides access to over 1000+ customer-facing drawings with customizable options for viewing various setscategories.
Single-use bioprocess systems reduce manufacturing costs byeliminatingthe need for complex cleaning, sterilization, and validation procedures associated with traditional stainless-steel equipment. Disposable components such astubing sets, sterile connectors, filtration systems, and fluid transfer assembliescan be installed quickly and discarded after use, minimizing downtime between production batches. This approach significantly reduces water, energy, and labor costs while improving operational efficiency. In addition, single-use systems allow manufacturers to scale production quickly and adapt to new therapeutic products without major infrastructure investments.
FoxxBioprocessproducts are widely used across industries that require sterile fluid handling and contamination-free workflows. These includebiotechnology, pharmaceutical manufacturing, vaccine development, clinical research laboratories, and life sciences research institutions. Single-use systems, filtration components, and bottle assemblies from Foxx help these organizationsmaintainproduct integrity and regulatory compliance while improving operational efficiency in both research and production environments.
When selecting a bioprocess equipment supplier, organizations should evaluate several key factors includingproduct quality, regulatory compliance, customization capabilities, and global supply reliability. The supplier should offer solutions designed for sterile and contamination-free processes, particularly for applications inbiologicsmanufacturing and pharmaceutical production. It is also important to ensure the products meetindustry standards such as ISO manufacturing practices and biocompatibility requirements. A good supplier will also providetechnical support, documentation, and flexible product configurationsto help laboratories and manufacturing facilitiesoptimizetheir bioprocess workflows.
Foxx Life Sciences is one of theLeading manufacturers of single-use bioprocess systemsanddevelop solutions designed for sterile fluid management, biologics production, and pharmaceutical manufacturing.Foxxprovidessingle-use bags, tubing assemblies, sterile connectors, filters, and fluid transfer systemsused in upstream and downstream processing. When selecting a manufacturer, companies typically considerquality certifications, material compatibility, supply chain reliability, and regulatory documentation. Established manufacturers also offercustom bioprocess assemblies and scalable solutionsthat support research laboratories, biotechnology companies, and large biopharmaceutical production facilities.
FoxxBioprocessprovides a comprehensive portfolio ofbioprocess components and laboratory solutionsdesigned for sterile fluid handling. Key products includesingle-use bottle assemblies, carboys, tubing systems, sterile connectors, filtration products, and custom fluid management assemblies. Many Foxx systems are built around technologies such asVersaCap® leak-proof caps andEZBio® single-use assembliesthat enable flexible and secure fluid transfer. These solutions are widely used in biotechnology laboratories, pharmaceutical production facilities, and research institutionsrequiringsterile fluid management systems.
Single-use bioprocess equipment can be sourced from specialized manufacturers and suppliers that focus on biotechnology and pharmaceutical manufacturing solutions. Reliable supplierslike Foxx LifeSciences,provide products such asbioprocess tubing assemblies, sterile connectors, filtration systems, bottle assemblies, and fluid transfer componentsdesigned for sterile and contamination-free operations. When choosing a supplier, it is important to evaluate factors likeproduct quality, regulatory compliance, manufacturing standards, customization capabilities, and supply reliability. A trusted bioprocess supplier can help laboratories, contract manufacturing organizations, and pharmaceutical companies implement efficient single-use workflows that improve productivity and support scalablebiologicsproduction.
Single-use bioprocessing technology uses disposable components such asbioprocess bags, tubing assemblies, connectors, bottle systems, and filtersthat are used once and discarded. This approach reduces contamination risks andeliminatescleaning and sterilization stepsrequiredin traditional stainless-steel systems. FoxxBioprocessoffers a wide range ofsingle-use assemblies including bottle assemblies, centrifuge tube assemblies, and fluid transfer systemsthat help biotech and pharmaceutical companies increase manufacturing flexibility and efficiency. These systems are widely used inbiologicsproduction and cell culture workflows.
Bioprocessing is the use of biological systems such as cells or micro-organisms to produce products like vaccines, biologics, and therapeutic proteins. FoxxBioprocesssupports these workflows through a comprehensive range ofsingle-use bioprocess systems, tubing assemblies, connectors, bottle systems, and filtration components. These solutions enable sterile fluid transfer, media preparation, and sample handling in laboratory and production environments. Foxx Life Sciences’EZBio® product line and single-use assembliesare designed to simplify upstream and downstream processes whilemaintainingsterility and regulatory compliance in biotechnology and pharmaceutical manufacturing.


Thanks for subscribing!
This email has been registered!
| Product | SKU | Description | Collection | Availability | Product Type | Other Details |
|---|